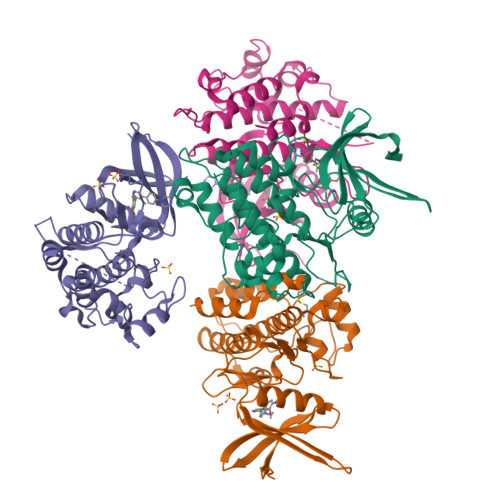
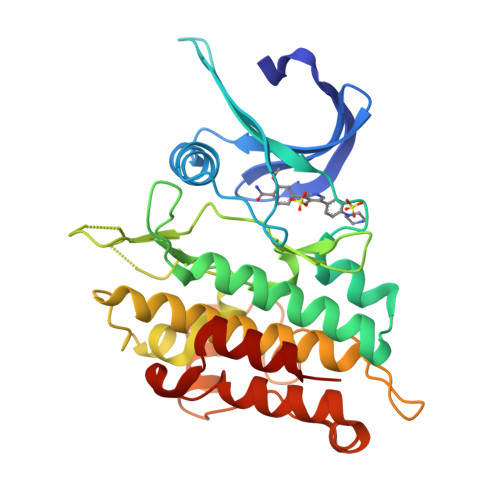
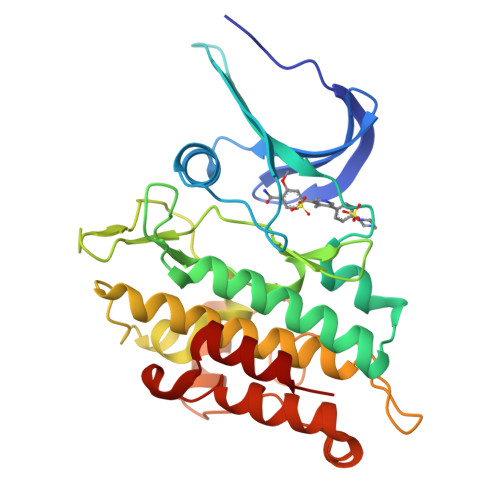
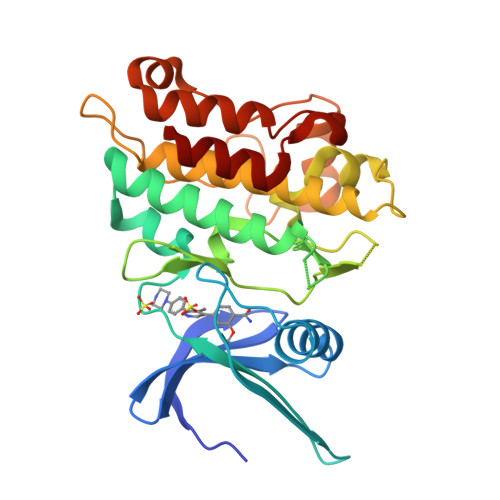
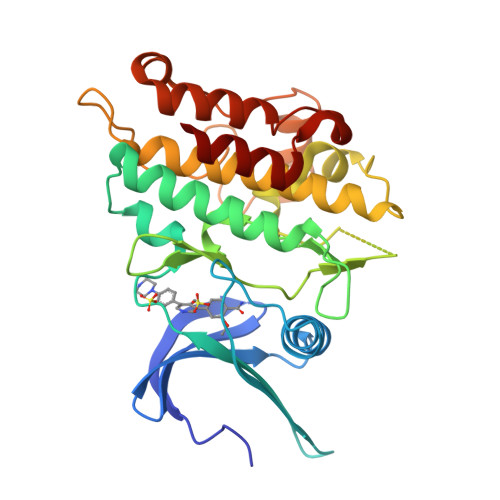
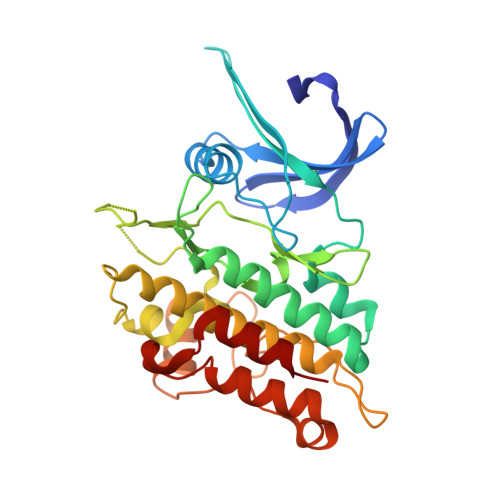
Targeting ALK2: An Open Science Approach to Developing Therapeutics for the Treatment of Diffuse Intrinsic Pontine Glioma.
Ensan, D., Smil, D., Zepeda-Velazquez, C.A., Panagopoulos, D., Wong, J.F., Williams, E.P., Adamson, R., Bullock, A.N., Kiyota, T., Aman, A., Roberts, O.G., Edwards, A.M., O'Meara, J.A., Isaac, M.B., Al-Awar, R.(2020) J Med Chem 63: 4978-4996
- PubMed: 32369358
- DOI: https://doi.org/10.1021/acs.jmedchem.0c00395
- Primary Citation of Related Structures:
6T6D, 8R7G - PubMed Abstract:
Diffuse intrinsic pontine glioma is an aggressive pediatric cancer for which no effective chemotherapeutic drugs exist. Analysis of the genomic landscape of this disease has led to the identification of the serine/threonine kinase ALK2 as a potential target for therapeutic intervention. In this work, we adopted an open science approach to develop a series of potent type I inhibitors of ALK2 which are orally bio-available and brain-penetrant. Initial efforts resulted in the discovery of M4K2009 , an analogue of the previously reported ALK2 inhibitor LDN-214117 . Although highly selective for ALK2 over the TGF-βR1 receptor ALK5, M4K2009 is also moderately active against the hERG potassium channel. Varying the substituents of the trimethoxyphenyl moiety gave rise to an equipotent benzamide analogue M4K2149 with reduced off-target affinity for the ion channel. Additional modifications yielded 2-fluoro-6-methoxybenzamide derivatives ( 26a-c ), which possess high inhibitory activity against ALK2, excellent selectivity, and superior pharmacokinetic profiles.
Organizational Affiliation:
Department of Pharmacology and Toxicology, University of Toronto, Medical Sciences Building, Room 4207, 1 King's College Circle, Toronto, Ontario M5S 1A8, Canada.